Type 2 diabetes mellitus
Adult: 5 mg once daily.
|
Indications and Dosage
Oral
Type 2 diabetes mellitus Adult: 5 mg once daily.
|
|
Administration
May be taken with or without food.
|
|
Special Precautions
Patient w/ history of pancreatitis; angioedema to other DPP-4 inhibitor. Not intended in patients w/ IDDM or for the treatment of diabetic ketoacidosis. Pregnancy and lactation.
|
|
Adverse Reactions
Hypoglycaemia, severe and disabling arthralgia, nasopharyngitis, rash, mouth ulcer, stomatitis, diarrhoea, cough. Rarely, hypersensitivity reactions.
Potentially Fatal: Acute pancreatitis. |
|
Monitoring Parameters
Monitor HbA1C and serum glucose levels; signs/symptoms of pancreatitis.
|
|
Drug Interactions
Increased risk of hypoglycaemia when used w/ an insulin secretagogue (e.g. sulfonylurea) or insulin. Plasma concentration of linagliptin may be decreased by strong inducers of P-glycoprotein (e.g. rifampicin) and may be increased by strong P-glycoprotein inhibitors (e.g. ritonavir).
|
|
Action
Description: Linagliptin inhibits dipeptidyl peptidase-4 (DPP-4), an enzyme which is involved in the inactivation of the incretin hormones GLP-1 (glucagon-like peptide-1) and GIP (glucose-dependent insulinotropic polypeptide). Both incretin hormones are involved in the physiological regulation of glucose homeostasis. Inhibition of DPP-4 leads to increased and prolonged active incretin levels.
Pharmacokinetics: Absorption: Rapidly absorbed from the GI tract. Bioavailability: Approx 30%. Time to peak plasma concentration: W/in approx 1.5 hr. Distribution: Extensively distributed to body tissues. Plasma protein binding: Concentration dependent: 99% (low concentration); 70-80% (high concentration). Metabolism: Not extensively metabolised. Excretion: Via faeces (approx 80%) and urine (5%). Terminal half-life: >100 hr. |
|
Chemical Structure
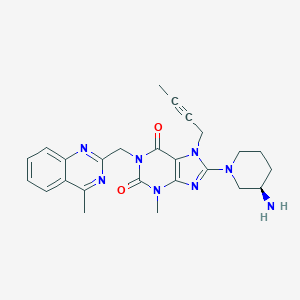 Source: National Center for Biotechnology Information. PubChem Database. Linagliptin, CID=10096344, https://pubchem.ncbi.nlm.nih.gov/compound/Linagliptin (accessed on Jan. 22, 2020) |
|
Storage
Store at 25°C.
|
|
MIMS Class
|
|
ATC Classification
A10BH05 - linagliptin ; Belongs to the class of dipeptidyl peptidase 4 (DPP-4) inhibitors. Used in the treatment of diabetes.
|
|
References
Anon. Linagliptin. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 24/09/2015 . Buckingham R (ed). Linagliptin. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 24/09/2015. McEvoy GK, Snow EK, Miller J et al (eds). Linagliptin. AHFS Drug Information (AHFS DI) [online]. American Society of Health-System Pharmacists (ASHP). https://www.medicinescomplete.com. Accessed 24/09/2015 . Tradjenta Tablet, Film Coated (Boehringer Ingelheim Pharmaceuticals, Inc.). DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 24/09/2015 .
|