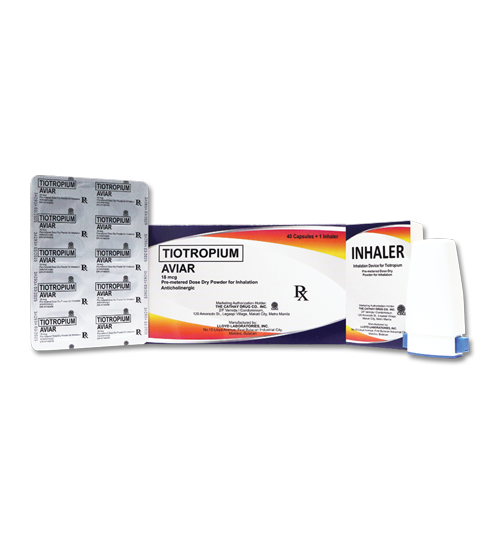
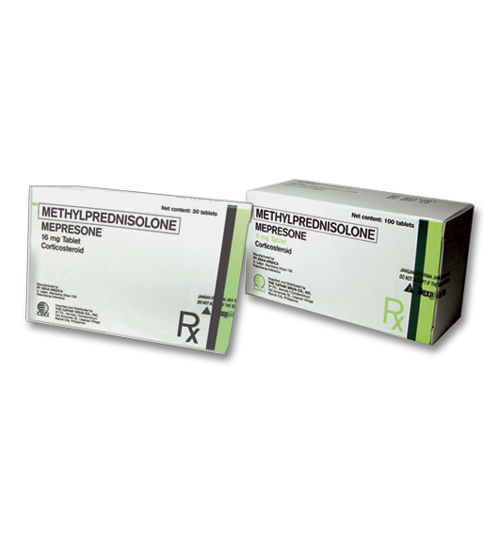
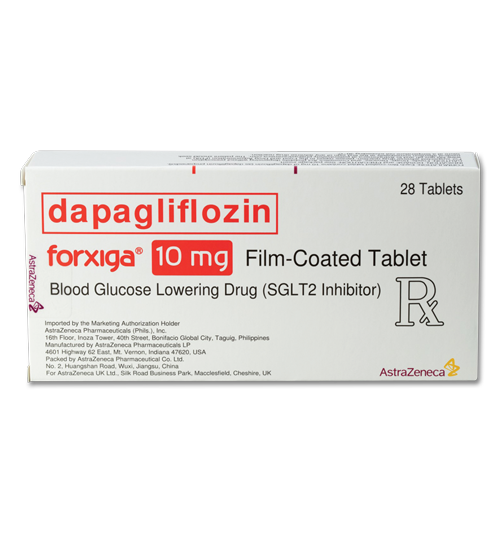
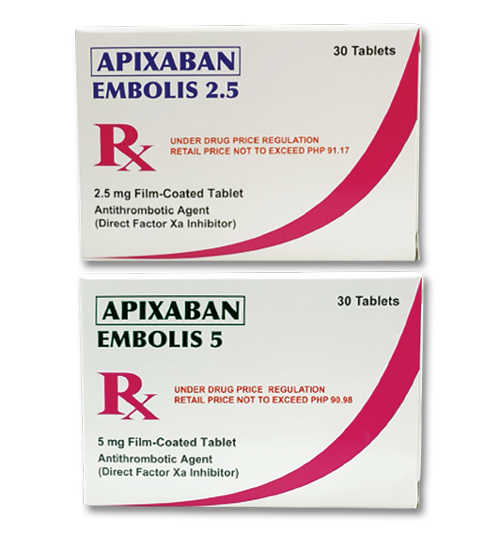
- education |
- points
- | UnregisterUser
- | IsNClickFreeLimitExceeded: False
- | NClickFreeLimit: 0
- |


UserName
Profession Email
Profession Email


 Sign Out
Sign Out