Type 2 diabetes mellitus
Adult: 60 or 120 mg tid. Max: 180 mg tid.
|
Indications and Dosage
Oral
Type 2 diabetes mellitus Adult: 60 or 120 mg tid. Max: 180 mg tid.
|
|
Administration
Should be taken with food. Take immediately before or up to 30 min before meals.
|
|
Contraindications
IDDM, diabetic ketoacidosis.
|
|
Special Precautions
Patient w/ adrenal and/or pituitary impairment. Severe renal and moderate to severe hepatic impairment. Pregnancy and lactation.
|
|
Adverse Reactions
Hypoglycaemia, upper resp tract infection, back pain, flu-like symptoms, dizziness, arthropathy, diarrhoea, accidental trauma, bronchitis, cough.
|
|
Monitoring Parameters
Monitor wt, lipid profile; fasting blood glucose and glycosylated Hb (HbA1c) levels.
|
|
Overdosage
Symptoms: Hypoglycaemia. Management: Use IV glucose in severe reaction.
|
|
Drug Interactions
CYP2C9 and CYP3A4 inhibitors or inducers may alter metabolism of nateglinide. Increased hypoglycaemic effects w/ MAOIs, nonselective β-adrenergic blockers, NSAIDs, salicylates. Decreased hypoglycaemic effects w/ corticosteroids, sympathomimetic agents, thiazide diuretics, thyroid hormones.
|
|
Food Interaction
Delayed absorption w/ food. Decreased serum concentration w/ St John's wort. Increased hypoglycaemic effects w/ alcohol.
|
|
Action
Description: Nateglinide, a nonsulfonylurea hypoglycaemic agent which stimulates insulin release from the pancreatic β-cells by blocking ATP-dependent K channels, depolarising the membrane and facilitating Ca entry through Ca channels. This action depends on the amount of existing glucose levels.
Onset: Approx 20 min. Duration: 4 hr. Pharmacokinetics: Absorption: Rapidly absorbed from the GI tract. Food delays absorption. Bioavailability: 73%. Time to peak plasma concentration: W/in 1 hr. Distribution: Volume of distribution: 10 L. Plasma protein-binding: 98%, primarily to albumin. Metabolism: Hepatic metabolism via hydroxylation followed by glucuronide conjugation by CYP2C9 and CYP3A4 (lesser extent) isoenzymes. Excretion: Mainly via urine (as unchanged drug and metabolites) and faeces (approx 10%). Elimination half-life: Approx 1.5 hr. |
|
Chemical Structure
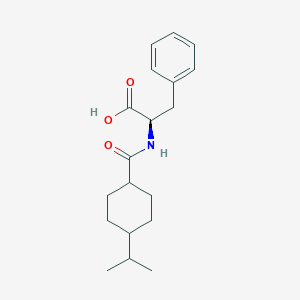 Source: National Center for Biotechnology Information. PubChem Database. Nateglinide, CID=5311309, https://pubchem.ncbi.nlm.nih.gov/compound/Nateglinide (accessed on Jan. 22, 2020) |
|
Storage
Store at 25°C.
|
|
MIMS Class
|
|
ATC Classification
A10BX03 - nateglinide ; Belongs to the class of other blood glucose lowering drugs, excluding insulins. Used in the treatment of diabetes.
|
|
References
Anon. Nateglinide. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 14/10/2015. Buckingham R (ed). Nateglinide. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 14/10/2015. McEvoy GK, Snow EK, Miller J et al (eds). Nateglinide. AHFS Drug Information (AHFS DI) [online]. American Society of Health-System Pharmacists (ASHP). https://www.medicinescomplete.com. Accessed 14/10/2015. Nateglinide Tablet (Dr. Reddy’s Laboratories Limited) . DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 14/10/2015.
|