Benign prostatic hyperplasia
Adult: 500 mcg once daily for at least 6 mth.
Child: Contraindicated.
Child: Contraindicated.
|
Indications and Dosage
Oral
Benign prostatic hyperplasia Adult: 500 mcg once daily for at least 6 mth.
Child: Contraindicated. |
|
Hepatic Impairment
Severe: Contraindicated.
|
|
Administration
May be taken with or without food.
|
|
Contraindications
Women, childn and adolescents. Severe hepatic impairment. Pregnancy and lactation.
|
|
Special Precautions
Mild to moderate hepatic impairment.
|
|
Adverse Reactions
Impotence, decreased libido, ejaculation disorder, breast disorders (e.g. tenderness, enlargement), breast cancer, hypersensitivity reactions (e.g. rash, pruritus, urticaria, localised oedema, serious skin reactions, angioedema), depressed mood, testicular pain and swelling, decreased prostate-specific antigen (PSA) concentration.
|
|
Patient Counseling Information
Avoid donating blood during treatment and for at least 6 mth following treatment cessation.
|
|
Monitoring Parameters
Monitor prostate-specific antigen (PSA) levels.
|
|
Drug Interactions
Plasma concentrations may be increased when used concomitantly w/ moderate or potent CYP3A4 inhibitors.
|
|
Lab Interference
May interfere with interpretation of prostate-specific antigen test results.
|
|
Action
Description: Dutasteride, a 4-azo analog of testosterone, is a selective inhibitor of steroid 5α-reductase isoenzymes type 1 (skin and hepatic) and type 2 (reproductive tissues). This results in inhibition of the conversion of testosterone to dihydrotestosterone and markedly suppresses serum dihydrotestosterone levels.
Pharmacokinetics: Absorption: Absorbed from the GI tract. Bioavailability: Approx 60%. Time to peak plasma concentration: 1-3 hr. Distribution: Plasma protein binding: >99.5%. Metabolism: Undergoes hepatic metabolism by CYP3A4 and CYP3A5 isoenzymes. Excretion: Via faeces (40% as metabolites; approx 5% as unchanged drug) and urine (<1% as unchanged drug). Elimination half-life: Approx 3-5 wk. |
|
Chemical Structure
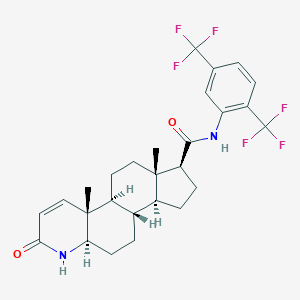 Source: National Center for Biotechnology Information. PubChem Database. Dutasteride, CID=6918296, https://pubchem.ncbi.nlm.nih.gov/compound/Dutasteride (accessed on Jan. 21, 2020) |
|
Storage
Store at 25°C.
|
|
MIMS Class
|
|
References
Anon. Dutasteride. AHFS Clinical Drug Information [online]. Bethesda, MD. American Society of Health-System Pharmacists, Inc. https://www.ahfscdi.com. Accessed 12/11/2014. Anon. Dutasteride. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 12/11/2014. Avodart- Dutasteride Capsule, Liquid filled. DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 12/11/2014. Buckingham R (ed). Dutasteride. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 12/11/2014.
|