Ocular hypertension, Open-angle glaucoma
Adult: As 2% soln: Instill 1 drop tid; bid when used w/ a topical β-blocker.
|
Indications and Dosage
Ophthalmic
Ocular hypertension, Open-angle glaucoma Adult: As 2% soln: Instill 1 drop tid; bid when used w/ a topical β-blocker.
|
|
Contraindications
Hyperchloraemic acidosis.
|
|
Special Precautions
Patient w/ history of renal calculi, pre-existing chronic corneal defects and/or history of intra-ocular surgery. Hepatic and severe renal (CrCl <30 mL/min) impairment. Pregnancy and lactation.
|
|
Adverse Reactions
Ocular burning, stinging and discomfort; superficial punctate keratitis, ocular allergic reactions (e.g. conjunctivitis, itching, lid reactions), blurred vision, redness, tearing, dryness, photophobia, transient myopia, eyelid crusting, palpebral reactions, ocular pain, irreversible corneal decompensation, ocular hypotony; acidosis, electrolyte imbalance, systemic allergic reactions (e.g. angioedema, pruritus, bronchospasm, urticaria), dizziness, paraesthesia, throat irritation, dyspnoea, contact dermatitis, bitter taste, headache, nausea, asthenia/fatigue, urolithiasis. Rarely, iridocyclitis.
Potentially Fatal: Stevens-Johnson syndrome, toxic epidermal necrolysis, fulminant hepatic necrosis, agranulocytosis, aplastic anaemia, and other blood dyscrasias. |
|
Ophth: C
|
|
Patient Counseling Information
This drug may cause dizziness and visual disturbances, if affected do not drive and operate machinery.
|
|
Monitoring Parameters
Monitor intraocular pressure periodically.
|
|
Drug Interactions
May increase the serum concentration of α-/β-agonists (indirect-acting). May enhance the adverse/toxic effects of other carbonic anhydrase inhibitors.
|
|
Action
Description: Dorzolamide reversibly inhibits carbonic anhydrase resulting in reduction of hydrogen ion secretion at renal tubule and increased renal excretion of Na, K, bicarbonate, and water, thus decreasing the production of aqueous humor. It also inhibits carbonic anhydrase in CNS to retard abnormal and excessive discharge from CNS neurons.
Duration: 8-12 hr. Pharmacokinetics: Absorption: Systemically absorbed. Distribution: Accumulates in RBC during chronic admin. Plasma protein binding: Approx 33%. Metabolism: Converted to N-desethyl metabolite. Excretion: Via urine, as unchanged drug and metabolites. |
|
Chemical Structure
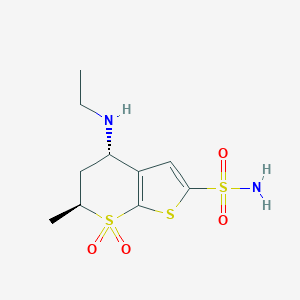 Source: National Center for Biotechnology Information. PubChem Database. Dorzolamide, CID=5284549, https://pubchem.ncbi.nlm.nih.gov/compound/Dorzolamide (accessed on Jan. 21, 2020) |
|
Storage
Store between 15-30°C. Protect from light.
|
|
MIMS Class
|
|
References
Anon. Dorzolamide. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 02/10/2014. Buckingham R (ed). Dorzolamide Hydrochloride. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 02/10/2014. Dorzolamide HCl Solution/Drops (Bausch & Lomb Incorporated). DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 02/10/2014. McEvoy GK, Snow EK, Miller J et al (eds). Dorzolamide Hydrochloride (EENT). AHFS Drug Information (AHFS DI) [online]. American Society of Health-System Pharmacists (ASHP). https://www.medicinescomplete.com. Accessed 02/10/2014.
|