Tinea pedis
Adult: As 2% cream: Apply to affected and immediate surrounding areas bid for 4 weeks.
|
Indications and Dosage
Topical/Cutaneous
Tinea pedis Adult: As 2% cream: Apply to affected and immediate surrounding areas bid for 4 weeks.
|
|
Contraindications
Hypersensitivity.
|
|
Special Precautions
Discontinue if irritation or sensitivity develop. Pregnancy and lactation.
|
|
Adverse Reactions
Contact dermatitis, dry skin, burning skin, application site reaction, skin tenderness, erythema, pruritus, vesiculation, desquamation, hyperpigmentation.
|
|
Topical: C
|
|
Patient Counseling Information
Prior to initiation of therapy, confirm diagnosis using KOH or cultures.
|
|
Monitoring Parameters
Reassess diagnosis if no clinical improvement after 2 wk.
|
|
Action
Description: Sertaconazole alters fungal cell wall membrane permeability. It inhibits the CYP450-dependent synthesis of ergosterol.
Pharmacokinetics: Absorption: Minimal (topical). |
|
Chemical Structure
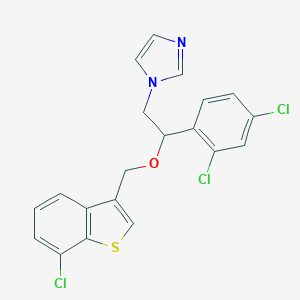 Source: National Center for Biotechnology Information. PubChem Database. Sertaconazole, CID=65863, https://pubchem.ncbi.nlm.nih.gov/compound/Sertaconazole (accessed on Jan. 23, 2020) |
|
Storage
Store between 20-25°C.
|
|
References
Anon. Sertaconazole. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 15/07/2014. Buckingham R (ed). Sertaconazole Nitrate. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 15/07/2014. Ertaczo (sertaconazole nitrate) cream (Valeant Pharmaceuticals North America LLC). DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed/. Accessed 15/07/2014. Ertaczo. U.S. FDA. https://www.fda.gov/. Accessed 15/07/2014. McEvoy GK, Snow EK, Miller J et al (eds). Sertaconazole Nitrate (Topical). AHFS Drug Information (AHFS DI) [online]. American Society of Health-System Pharmacists (ASHP). https://www.medicinescomplete.com. Accessed 15/07/2014. Wickersham RM. Sertaconazole Nitrate - Topical. Facts and Comparisons [online]. St. Louis, MO. Wolters Kluwer Clinical Drug Information, Inc. https://www.wolterskluwercdi.com/facts-comparisons-online/. Accessed 15/07/2014.
|