Hypertension
Adult: Initially, 10 mg once daily, may increase to 20 mg once daily after at least 2 weeks according to patient's response.
|
Indications and Dosage
Oral
Hypertension Adult: Initially, 10 mg once daily, may increase to 20 mg once daily after at least 2 weeks according to patient's response.
|
||||
|
Renal Impairment
|
||||
|
Hepatic Impairment
Severe: Contraindicated.
|
||||
|
Administration
Should be taken on an empty stomach. Take at least 15 min before meals.
|
||||
|
Contraindications
Left ventricular outflow obstruction (aortic stenosis), untreated CHF, unstable angina, recent MI (within 1 month). Severe hepatic and renal impairment (CrCl <30 mL/min). Pregnancy and lactation. Concomitant use with strong CYP3A4 inhibitors, and cyclosporin.
|
||||
|
Special Precautions
Patient with sick sinus syndrome without a functioning pacemaker, left ventricular dysfunction, ischaemic heart disease. Mild to moderate renal and hepatic impairment.
|
||||
|
Adverse Reactions
Significant: Angina pectoris, hypotension.
Cardiac disorders: Tachycardia, palpitations. Gastrointestinal disorders: Dyspepsia, nausea, upper abdominal pain. General disorders and admin site conditions: Peripheral oedema, asthenia, fatigue. Musculoskeletal and connective tissue disorders: Myalgia. Nervous system disorders: Headache, dizziness. Renal and urinary disorders: Polyuria. Skin and subcutaneous tissue disorders: Rash, pruritus. Vascular disorders: Flushing. |
||||
|
Patient Counseling Information
This drug may cause dizziness, weakness and fatigue, if affected, do not drive or operate machinery.
|
||||
|
Monitoring Parameters
Monitor heart rate and blood pressure.
|
||||
|
Overdosage
Symptoms: Peripheral vasodilation, hypotension, tachycardia, bradycardia and negative inotropic effect (at very high doses), dizziness, headache and palpitations. Management: Supportive treatment. Monitor cardiac and respiratory function, circulating fluid volume, and urine output.
|
||||
|
Drug Interactions
Reduced antihypertensive effect with CYP3A4 inducers (e.g. phenytoin, rifampicin). Increased absorption with midazolam. Decreased serum concentration with metoprolol.
Potentially Fatal: Increased serum concentration with ciclosporin and strong CYP3A4 inhibitors (e.g. ketoconazole, ritonavir, erythromycin). |
||||
|
Food Interaction
Enhanced vasodilatory effect with alcohol. Increased serum concentration with grapefruit juice.
|
||||
|
Action
Description: Lercanidipine is a dihydropyridine calcium antagonist which selectively inhibits the transmembrane influx of Ca into cardiac and vascular smooth muscle (greater effect on vascular than on cardiac smooth muscle) during depolarisation. It reduces peripheral vascular resistance which leads to lower arterial blood pressure by directly relaxing the vascular smooth muscle.
Duration: 24 hours. Pharmacokinetics: Absorption: Rapidly and completely absorbed from the gastrointestinal tract. Bioavailability: Approx 10%. Time to peak plasma concentration: Approx 1.5-3 hours. Distribution: Rapidly and widely distributed to tissues and organs. Volume of distribution: 2 to 2.5 L/kg. Plasma protein binding: >98%. Metabolism: Extensively metabolised by CYP3A4 into inactive metabolites; undergoes extensive first-pass metabolism. Excretion: Via urine (approx 50% as metabolites). Elimination half-life: 8-10 hours. |
||||
|
Chemical Structure
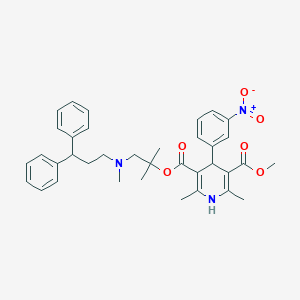 Source: National Center for Biotechnology Information. PubChem Database. Lercanidipine, CID=65866, https://pubchem.ncbi.nlm.nih.gov/compound/Lercanidipine (accessed on Jan. 22, 2020) |
||||
|
Storage
Store below 30°C. Protect from light and moisture.
|
||||
|
MIMS Class
|
||||
|
ATC Classification
C08CA13 - lercanidipine ; Belongs to the class of dihydropyridine derivative selective calcium-channel blockers with mainly vascular effects. Used in the treatment of cardiovascular diseases.
|
||||
|
References
Anon. Lercanidipine. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 18/05/2021. Buckingham R (ed). Lercanidipine Hydrochloride. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 18/05/2021. Zanidip 20 mg Tablets (Recordati Industria Chimica e Farmaceutica S.p.A). MHRA. https://products.mhra.gov.uk. Accessed 18/05/2021. Zanidip Film-Coated Tablets (Recordati Industria Chimica e Farmaceutica S.p.A). National Pharmaceutical Regulatory Agency - Ministry of Health Malaysia. https://www.npra.gov.my. Accessed 18/05/2021.
|