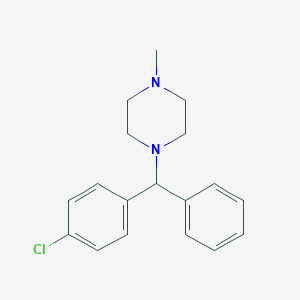
Allergic conditions
Adult: 25 mg 6-8 hrly. Max: 75 mg 24 hrly.
Child: 6-11 yr 12.5 mg 6-8 hrly. Max: 37.5 mg 24 hrly; ≥12 yr Same as adult dose.
Child: 6-11 yr 12.5 mg 6-8 hrly. Max: 37.5 mg 24 hrly; ≥12 yr Same as adult dose.
Oral
Motion sickness
Child: ≥6 yr As chewable tab: 25 mg 6-8 hrly taken up to 1 hr before travel or at onset of symptoms.




 Sign Out
Sign Out