Benign prostatic hyperplasia
Adult: As monotherapy or in combination with α-1 adrenergic antagonist: 0.5 mg once daily. Assess treatment after at least 6 months; treatment duration depends on patient’s clinical response.
|
Indications and Dosage
Oral
Benign prostatic hyperplasia Adult: As monotherapy or in combination with α-1 adrenergic antagonist: 0.5 mg once daily. Assess treatment after at least 6 months; treatment duration depends on patient’s clinical response.
|
|
Hepatic Impairment
Severe: Contraindicated.
|
|
Administration
May be taken with or without food.
|
|
Contraindications
Severe hepatic impairment. Women, children and adolescents. Pregnancy and lactation.
|
|
Special Precautions
Patient with large post-void residual urine volume or severely reduced urinary flow. Mild to moderate hepatic impairment. Not indicated for the prevention of prostate cancer.
|
|
Adverse Reactions
Significant: Cardiac failure; reduction in total sperm count, semen volume, and sperm motility. Rarely, breast cancer.
Immune system disorders: Allergic reactions (e.g. rash, pruritus, urticaria, localised oedema, angioedema). Investigations: Increased LH, testosterone, TSH. Nervous system disorders: Dizziness. Psychiatric disorders: Depression. Reproductive system and breast disorders: Testicular pain and swelling, impotence, decreased libido, ejaculation disorders, breast disorders (e.g. tenderness, enlargement). Skin and subcutaneous tissue disorders: Alopecia (primarily body hair loss), hypertrichosis. |
|
Patient Counseling Information
Avoid donating blood during treatment and for at least 6 months following treatment cessation. Women of child-bearing potential or who are pregnant should not handle crushed or broken tab. Excreted in semen therefore use of condom is recommended.
|
|
Monitoring Parameters
Evaluate patients for prostate cancer (e.g. digital rectal examinations) prior to treatment and periodically thereafter. Monitor prostate-specific antigen (PSA) levels (new baseline after 6 months from treatment initiation then regularly thereafter); urinalysis (baseline). Assess for signs and symptoms of changes in breast tissue (e.g. pain, nipple discharge, lumps).
|
|
Drug Interactions
Increased serum concentrations with moderate CYP3A4 inhibitors (e.g. verapamil, diltiazem), or potent CYP3A4 inhibitors (e.g. ritonavir, indinavir, nefazodone, itraconazole, ketoconazole).
|
|
Lab Interference
Decrease PSA level by approx 50% within 3-6 months of use.
|
|
Action
Description:
Mechanism of Action: Dutasteride, a 4-azo analog of testosterone, is a competitive, selective inhibitor of both type 1 (skin and liver) and type 2 (reproductive tissues) 5α-reductase, resulting in the inhibition of the conversion of testosterone to dihydrotestosterone thus reducing levels of circulating dihydrotestosterone. Pharmacokinetics: Absorption: Absorbed from the gastrointestinal tract. Bioavailability: Approx 60%. Time to peak plasma concentration: 1-3 hours. Distribution: Volume of distribution: 300-500 L. Plasma protein binding: 99% to albumin. Metabolism: Extensively metabolised in the liver by CYP3A4 and CYP3A5 isoenzymes into 6-hydroxydutasteride (same activity with dutasteride); 4′-hydroxydutasteride and 1,2-dihydrodutasteride (less potent than dutasteride). Excretion: Via faeces (40% as metabolites, approx 5% as unchanged drug); urine (<1% as unchanged drug). Elimination half-life: Approx 3-5 weeks. |
|
Chemical Structure
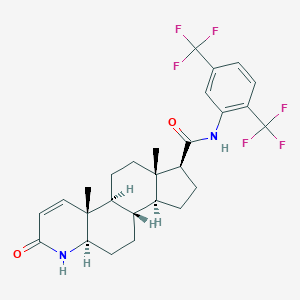 Source: National Center for Biotechnology Information. PubChem Compound Summary for CID 6918296, Dutasteride. https://pubchem.ncbi.nlm.nih.gov/compound/Dutasteride. Accessed Nov. 22, 2023. |
|
Storage
Store between 15-30°C. Follow applicable procedures for receiving, handling, administration, and disposal.
|
|
MIMS Class
|
|
ATC Classification
G04CB02 - dutasteride ; Belongs to the class of testosterone-5-alpha reductase inhibitors. Used in the treatment of benign prostatic hypertrophy.
|
|
References
Anon. Dutasteride. AHFS Clinical Drug Information [online]. Bethesda, MD. American Society of Health-System Pharmacists, Inc. https://www.ahfscdi.com. Accessed 25/09/2023. Anon. Dutasteride. Lexicomp Online. Hudson, Ohio. Wolters Kluwer Clinical Drug Information, Inc. https://online.lexi.com. Accessed 25/09/2023. Avodart 0.5 mg Capsule (GlaxoSmithKline Pharmaceutical Sdn. Bhd.). National Pharmaceutical Regulatory Agency - Ministry of Health Malaysia. https://www.npra.gov.my. Accessed 25/09/2023. Avodart Capsule, Liquid Filled (Woodward Pharma Services LLC). DailyMed. Source: U.S. National Library of Medicine. https://dailymed.nlm.nih.gov/dailymed. Accessed 25/09/2023. Buckingham R (ed). Dutasteride. Martindale: The Complete Drug Reference [online]. London. Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 25/09/2023. Dutasteride 0.5 mg Soft Capsules (Ennogen IP Ltd,). MHRA. https://products.mhra.gov.uk. Accessed 25/09/2023. Joint Formulary Committee. Dutasteride. British National Formulary [online]. London. BMJ Group and Pharmaceutical Press. https://www.medicinescomplete.com. Accessed 25/09/2023.
|